
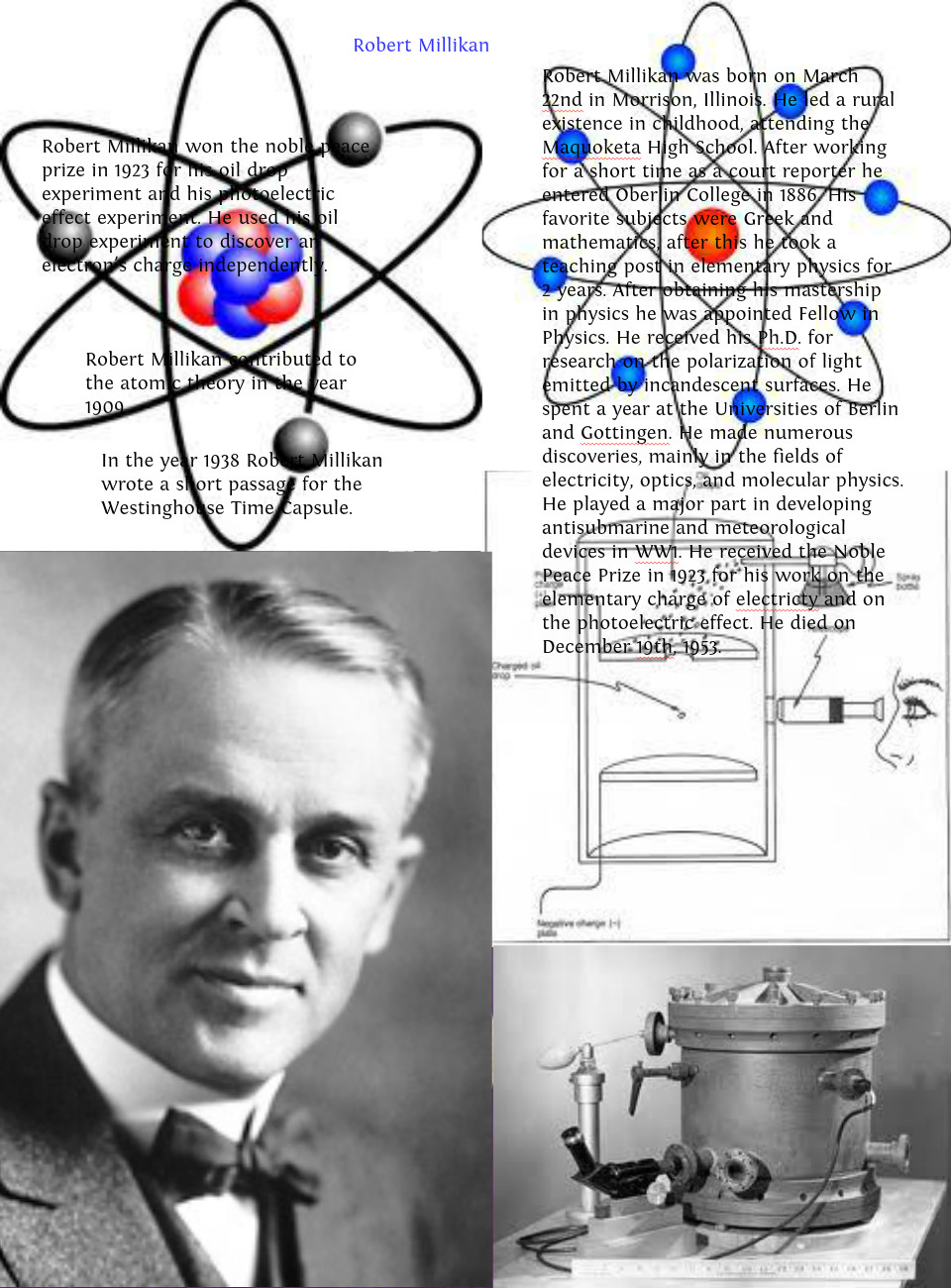


In these experiments, the atomizer from a perfume bottle was used to spray water or oil droplets into a sample chamber. Millikan The Man and his contributions to The Atomic Theory Robert a. Understand how his findings defined the law of conservation of mass. The Charge of an Electron (Robert Millikan) Between 19, Robert Millikan measured the charge on an electron with the apparatus shown below. The theory of the four elements was much more well-received. Member (elected 1915) Robert Millikan was a physicist who discovered the elementary charge of an electron using the oil-drop experiment. \)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles.\): Millikan’s experiment measured the charge of individual oil drops. Learn about Lavoisiers atomic theory and discovery. Atomic Theory: The Ancient Greeks proposed two basic theories about the elements, one which theorized there were four elements, earth, air, fire, and water, and the other that proposed that each substance was composed of small individual particles of that substance called atomies.
